Molecular Model of the Ring-Opening and Ring-Closure Reaction of a Fluorinated Indolylfulgide
10-Dec-2012
J. Phys. Chem. A, 2012, 116, 10518-10528 published on 29.09.2012
J.Phys.Chem.A
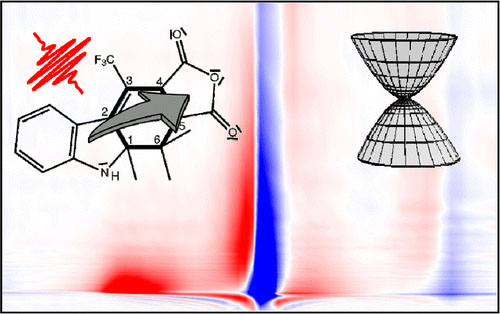
A combination of experimental and theoretical techniques is used to study the photoinduced ring-opening/closure of a trifluoromethyl-indolylfulgide. Time-resolved UV/vis pump and IR probe measurements are performed in the subpicosecond to 50 ps time range. A combination of experimental and theoretical techniques is used to study the photoinduced ring-opening/closure of a trifluoromethyl-indolylfulgide. Time-resolved UV/vis pump and IR probe measurements are performed in the subpicosecond to 50 ps time range. Probing in the mid-IR between 1200 and 1900 cm–1 provides mode-specific dynamics and reveals photochemical reaction dynamics as well as the presence of a noncyclizable conformer. Ring-opening occurs with about 3 ps. Experiments on the open isomer confirm that the ring-closure occurs on the subpicosecond time scale. They also show a 10 ps transient that can be assigned to internal conversion of a noncyclizable conformer. Quantum chemical calculations with multireference methods are used to explore the complex potential energy landscape in the excited electronic state showing paths for ring-opening and closure reactions as well as for competing side-reactions. The calculations reveal that photoexcitation induces a charge transfer from the indole to the anhydride. The same charge transfer drives the system toward a low-energy conical intersection seam spreading from the closed to the open ring side and is responsible for the ultrafast relaxation to the ground state. The ultrafast photoreactivity comes at the expense of selectivity.